KEY CONCEPTS
•
Lower-viscosity lubricants squeeze more fuel efficiency by reducing friction without compromising wear.
•
Less than 25% of the energy in a car’s fuel moves it along; of the rest most is lost to heat, with about 28% due to friction.
•
Both Group IV PAOs and Group V PAGs can improve fuel efficiency further by trimming molecular branching.
More than 35 years ago, the first oils emerged specifically designed to improve the fuel efficiency of vehicles, a standard known as ILSAC GF-1. Synthetic oils jumped in on this action—their improved stability and ability to work better at both low and high temperatures provided advantages that the mineral oil base stocks of the day could not.
Synthetics made from base stocks such as polyalphaolefins, or PAOs, improved fuel efficiency in a number of ways and provided better balance of properties over the mineral oils at the time. Now, with GF-6 standards a few years out, lubricant formulators reflect on the next steps to squeeze even greater gains in fuel efficiency from oils.
It won’t be simple, says TLT technical editor Reg Forbus, chief technology officer and president of the Lubricants Division at Santolubes, a synthetic base oil and lubricant manufacturer in Spartanburg, S.C.
“I believe we’ve pushed the envelope with PAOs and mineral oil processing about as far as we can in terms of obtaining much further gains in beneficial properties,” says Forbus. “I don’t think there are much further gains to be made with purely hydrocarbon-based base oils.”
One of the last big advances, according to STLE-member Lake Speed, Jr., head of R&D for Driven Racing Oil of Olive Branch, Miss., was in the early 2010s: chemists developed metallocene PAOs, also known as mPAOs, using a new way to synthesize the long chain hydrocarbon molecules.
“In the last 15 years, we’ve tested lots of different additives and base oils. The one that gave us the most immediate measurable benefit was the mPAOs, which is why I’m a pretty big advocate of them,” says Speed.
Speed first heard of mPAO while attending an STLE annual meeting where current STLE treasurer Ken Hope, a technical services manager at Chevron Phillips Chemical Co., presented new work with this base stock at the meeting. Speed recognized the advancement’s potential for use in racing cars (
1).
“Everything that he talked about was exactly what we were looking for in a base stock to help us move forward. We formulated some oils based on samples he sent us. We tested them, and we starting winning NASCAR races,” says Speed. “For our company that was incredibly exciting.”
Hope says there’s still room for improvement in PAOs.
“I haven’t seen an example of a petroleum-based, highly-isoparaffinic fluid that is equivalent to or better than PAO,” he says. “Oftentimes we see comparisons on the basis of a few properties, but we are finding that when you look at the balance of the overall properties, including the volatility, low-temperature viscometrics, oxidative stability, thermal conductivity and frictional properties, there is a different story. Targets are key. PAOs open up blending opportunities so formulators can tailor their products to meet specific technical targets.”
Forbus suggests that synthesized, optimally tailored, Group V base oils look the most promising for pushing the envelope further for base oil technologies and improved lubricant performance. PAOs are Group IV oils. Refined and processed mineral oils make up Groups I, II and III (
2).
One Group V synthetic is polyalkylene glycols, known as PAGs. PAGs are similar to hydrocarbon fluids but include oxygen in the backbone of the molecules. Forbus says that polyalkylene glycol base oils can be made with lower shear strength relative to purely hydrocarbon-based fluids. This property improvement contributes to higher energy efficiency in certain industrial oils, mainly in high-contact stress, high-sliding applications such as gear oils.
“You can produce PAGs that have good low-temperature properties, very high viscosity indexes and lower shear strength relative to hydrocarbon-based fluids. The caveat is the fact that in order to make PAGs that have all the properties required for lubricants, in terms of viscometrics, you have to put a lot of propylene into the molecules, which puts a lot of methyl branches on the backbone of those molecules,” Forbus says. “This actually limits the best potential properties of PAG molecules. But it’s a necessary evil to balance their properties for optimized lubricants.”
Turning down the heat
Many factors contribute to a vehicle’s energy inefficiency. Estimates say up to a quarter of the energy in a car’s fuel actually moves it along (
3). Most of the rest is lost to heat, with about almost a third of that loss due to friction. Some researchers predict friction losses could be cut about 18% in the next 10 years or so (
4).
Friction has many sources. Pistons are moving up and down inside the engine. The cam shaft is spinning. Gears and joints and pulleys are engaging and turning and slipping in the drivetrain. Lubricants can reduce friction of the internal parts to allow more of that heat energy to be put to work moving the vehicle.
Three factors will influence the energy efficiency of a lubricant, says STLE Life Member Vern Wedeven, president of Wedeven Associates in Edgemont, Penn.
First is how the fluid viscosity behaves with respect to temperature and pressure to build an oil film to accommodate shear without the resistance of roughness features. Also, how easily it shears, which is due to its internal friction, also known as the traction coefficient.
Second is the oil and additive chemistry, which influences the degree to which the height of roughness features are preserved or polished to cause traction. Third is the shear resistance of the boundary lubricating surface films that react with the surface chemistry of the contacting metallurgy.
“You have to understand the mechanisms behind all these properties, and then you can fine tune them to your application. No one oil can do everything,” says Wedeven. “An oil that someone says solves all your problems, we call that snake oil.”
Hope agreed that the applications will guide the next improvements. “Energy efficiency seems to have different drivers depending upon the lubrication application. For instance, improvements in efficiency for industrial lubricants and heavy-duty engine oils appear to be somewhat self-regulating, while passenger car fuel efficiencies are a bit more complex.”
Passenger cars, he points out, are subject to regulations that are different depending on where the cars will be driven, with the U.S. maintaining current average fuel economy standards, also known as CAFE, compared to other regions.
“There seems to be an evolving divergence in the regulatory directions of energy efficiency depending upon the region of the world. Europe and Asia are driving toward stricter or tighter regulations, while here in the U.S. there is a proposed leveling off from the 2020 CAFE limits,” Hope says.
Friction opportunities
Requirements and regulations might be a driving force, but the real forces formulators have to work with are those in the engine and drivetrains themselves in the form of friction.
Those sources of friction are the opportunities for efficiencies that concerns Wedeven. “In gearboxes or transmissions, part of the losses are due to gear meshing itself, but there are bearing and seal losses as well,” he says. “A lot of the losses also are the oil that’s just churning around in the system. The energy efficiency of the oil plays a role, but what are the interfaces that rub or slide or roll against each other? What causes the friction?”
Base oils with too much viscosity generate film thicknesses that actually create more friction and also higher consequential churning losses. How the thickness changes with temperature will determine how much protection the oils provide in cold and hot conditions, referred to as viscosity index. Oils with a high viscosity index maintain more similar viscosities at both low and high temperatures. Base oils refined from petroleum also have impurities that contribute to friction and wear, as well as the performance and aging of the oil itself (
5).
“We’re not using the base stocks from 40 years ago,” says Speed. “We’re using base stocks that’ve come out in the last 10 years. There’s not a single one in the family of conventional base stocks that perform as well as a synthetic—at least a polyalphaolefin or a metallocene polyalphaolefin. Those types of base oils are what are enabling higher efficiency without giving up wear.”
To make synthetic hydrocarbon base oils, manufacturers distill, break down, then reassemble the components to alkyl-branched long chain carbon molecules, derived typically from alphaolefins produced from ethylene or propylene. A PAO contains multiple alphaolefins linked together. Synthetic PAOs are more uniform than mineral oils but still have plenty of branches, similar to a bottle brush.
mPAOs are synthesized using a metallocene catalyst (
6). Creating molecules shaped more like a comb than a brush, this synthesis method results in more streamlined oil molecules that exhibit lower internal friction.
Formulators seek the least-viscous, thinnest oil that still protects surfaces moving against each other. Viscosity specifications of multi-grade oils such as 10W-40 include two terms that refer to two different scales. The first term, which includes “W” for winter, is the cold cranking ability, or how well the oil will flow below freezing temperatures.
“The lower the first number, the colder it can be outside and still safely start your engine,” says Speed.
The second number is a measure for flow at high temperatures. Again, the lower the number, the more easily it flows in the heat. Formulators’ goal is to provide just enough viscosity at the highest temperatures of operation to protect the rubbing surfaces for adequate protection from friction and wear.
“How do we make that next step in efficiency without compromising durability? That’s where reformulating in terms of what base stocks and additives will allow the industry to create even lower viscosity oil—say a 0W-12 that can provide better fuel efficiency than, say, a 5W-30 without compromising wear,” says Speed. “That’s the kind of work that myself, my company and others in industry are actively engaged in today.”
Because race car engines are limited in size, race oil formulators have to gain more power by making the engines more fuel efficient. “We’re able to prove at least in a test environment that we can go from a 5W-30 to a 0W-12 and increase fuel economy more than 5%, without compromising anything on wear. We do that by using advanced base stocks,” Speed says.
 Formulators for race car oils gain power by making the engines more fuel efficient.
Formulators for race car oils gain power by making the engines more fuel efficient.
© Can Stock Photo / Dave_Koontz
Branching out
Getting rid of branching in molecules, says Forbus, generally produces oil with lower shear strength, which is yet another term for traction coefficient. “You want to minimize lubricant’s shear strength in order to minimize the fluid’s required shearing forces and thereby maximize energy efficiency,” he says, “especially for lubrication associated with gears but also to some extent with rolling element bearings such as ball bearings, tapered roller bearings, cylindrical bearings, etc. The shearing of the lubricant constantly occurs between the gears in all gear teeth meshing cycles.”
In such contacts, known as elastohydrodynamic contacts, the lubrication solidifies. “And it’s the high-contact stress shear strength of the fluid as a glassy solid that becomes important. It’s the solid properties of the lubricant under those high-stress contact conditions,” says Forbus, “that governs the lubrication process and the energy requirement to shear the fluid in the contacts.”
Wedeven says the solidification happens due to high-contact stresses. “The amazing part and also the complex part is when gear teeth or bearing contacts are operating under enormous stress in the presence of slip. The pressure within oil initially captured in the contact causes the oil to go from a liquid to a pseudo-solid. And it shears like a solid with relatively high traction. As the oil transits the contact under stress and shear, it heats up and goes back to a viscous low-traction state.”
Understanding the details provides helpful insights, he says. “Solidification starts upstream of the contact to generate a film. Once captured, the shear-induced thermals reduce the internal friction or traction for efficiency. This amazing rheological behavior of the oil does the heavy lifting for film formation, as well as shear accommodation for efficiency. A good tribology design puts these oil mechanisms to work.”
Current PAG fluids are limited by the number of branching methyl groups required to balance their properties for good lubricants. Says Forbus: “So going forward, what’s necessary is to eliminate the level of branching in lubricant base oil molecules in order to push the envelope even higher in terms of their potential energy savings ability from lowering the shear strength of base oils. This, I believe, is where new Group V base oils offer the greatest opportunity.”
Going through a solid phase in a high-stress contact, Forbus says, is a necessary evil. “In order to create a lubricant film to minimize surface interactions in contacts, you have to form a film, Forbus says. “It just so happens that that film in concentrated contacts is a glassy solid momentarily in the contact, subject to plastic shear.
“Minimizing the shear strength of the ‘solidified’ fluid film relegates lower shearing forces and thereby reduces the required energy to shear the fluid in the contacts. This contributes to great energy efficiency in the lubrication process,” he adds.
Forbus’ takeaway is that substantial gains in the future will come from minimizing lubricant shear strength properties to make them shear easier within high-stress contacts and, thereby, expend less energy in the shearing process. This reflects directly on energy efficiency. Minimizing branching by making the base oil molecules more linear does this.
By addressing branching, Santolubes has created a base oil technology with very low shear strength, Forbus reports. “My company has created materials that only have about a third of the shear strength of typical PAOs, or equivalent very high-quality hydrocarbon fluids like Group III/III+ mineral oils. And only about half of the high stress shear strength of the best PAGs on the market (
see Figure 1).”
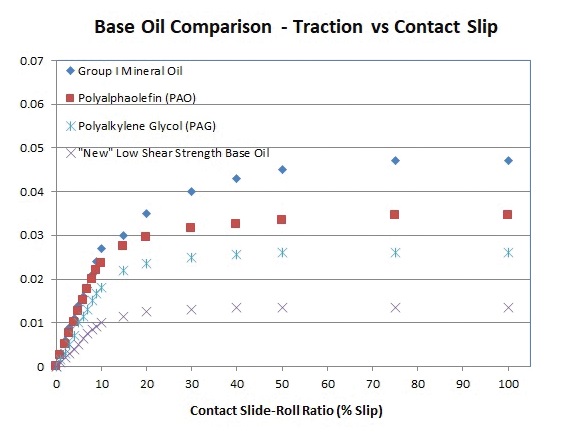 Figure 1. A recently developed base oil achieves very low shear strength compared to other general base oil types: mineral, PAO and PAG. (Figure courtesy of Santolubes.)
Figure 1. A recently developed base oil achieves very low shear strength compared to other general base oil types: mineral, PAO and PAG. (Figure courtesy of Santolubes.)
But it’s too early to know how that translates to energy savings. “Now that we have base oils that have substantially improved lower shear strength, we’re exploring just how much greater efficiency we can see in practice in real equipment. We are just now starting to scratch that surface,” says Forbus.
In addition to providing the right viscosities at different temperatures, synthetic lubricants must withstand high temperatures. Wedeven works on lubricants for gas turbines on jet engines. He says that contact surfaces on jet engine bearings and gears move at more than 100 miles an hour, with a portion of that motion under sliding, particularly for gears.
“Even at 100 mph, with 5% slip or so, we’re still dealing with enormously high-sliding velocities. That generates a lot of heat where the oil film can go over 300 C. It goes way up but only momentarily,” Wedeven says.
Formulating non-friction
Engine and gear oils are a combination of base oil and additives that, directly and indirectly, affect energy efficiency in several ways, including by keeping the engine running smoothly.
Additives have a variety of jobs. Detergents keep high-temperature areas like the piston ring lands clean. The dispersants suspend soot and prevent sludge from building up. ZDDP is an antioxidant that helps the oil stay alive by preventing age-induced oxidation, which breaks oil down.
“Once antioxidants are gone, the oil typically breaks down, and bad things happen if you keep running on that oil,” says Speed.
Anti-foam additives prevent oil from foaming up due to air—air, it turns out, makes a really bad lubricant. Rust inhibitors are important to prevent rust in the engine, especially now that ethanol-containing fuel is widespread. And friction modifiers affect how much energy is lost to heat while the lubricant lubricates.
“There are many different features in engine oil, especially compared to a gear oil or standard hydraulic fluid,” says Speed.
Whether energy efficiency gains will be the purview of the base oils or additives might depend on whether the base oil is polar or nonpolar.
Highly polar base oils, such as those in Group V, have higher inherent lubricity than nonpolar ones. Group IVs such as PAOs are nonpolar.
“Additives are there to compliment the lubricity that’s brought about by the base oil, so lubricity additives would be less important to the mix with Group Vs because the polarity of the base oil does a lot of the work. With polar base oils, the lubricity additive is playing a secondary role,” Speed adds.
Mountains and molehills
Surfaces also contribute to how much energy is lost to friction. In some applications, such as automobiles, the surfaces are relatively rough. But in the jet engine gas turbines that Wedeven works with, surfaces are very smooth, causing the oil films to be measured in microns or nanometers.
“Advancements in lubricant technology have to go hand-in-hand with materials and manufacturing methods that create surfaces that are accurate in geometry and roughness or topography,” Wedeven says. “Films that are generated for aerospace have to be very thin, stresses can be very high, sliding and rolling velocities can be very high, yet they can be run very efficiently for very long periods of time.”
Films get thinner as the temperature rises and, if they get thin enough, at some point roughness features on opposing surfaces can’t be fully separated at the interface—they start interacting with one another. To explore what happens then, Wedeven recently tested the intersection of chemistry and surfaces using analysis and simulation of a Humvee differential hypoid gear (
see Figure 2).
 Figure 2. Squeezing efficiency out of synthetics is about tuning properties to the application and its operation. For high-speed differential applications, viscous properties contributing to churning losses within the system are important. For low-speed high-torque applications, oil rheology properties to generate oil films to accommodate shear without friction from roughness features between hypoid gear teeth become important. Simulation testing and modeling of the tribology mechanisms in the contact are companion tools to formulate oil attributes for specific applications. (Figure courtesy of Wedeven Associates.)
Figure 2. Squeezing efficiency out of synthetics is about tuning properties to the application and its operation. For high-speed differential applications, viscous properties contributing to churning losses within the system are important. For low-speed high-torque applications, oil rheology properties to generate oil films to accommodate shear without friction from roughness features between hypoid gear teeth become important. Simulation testing and modeling of the tribology mechanisms in the contact are companion tools to formulate oil attributes for specific applications. (Figure courtesy of Wedeven Associates.)
“How do you make the chemistry so you not only protect the surface but make the surface smoother? That’s tricky business, but it can be done,” says Wedeven.
The idea, he says, is that the chemistry attaches to the surface only where there are high temperatures, high stresses and metal, where the chemistry can react to protect the surface. Almost like magic, the chemistry within the lubricant can both wear imperfections down and protect the underlying surface.
“You want the chemistry to allow the surface imperfections to wear down, but once they do you want the chemistry to prevent them from wearing further,” says Wedeven.
In a November 2018 (
7) publication, Wedeven and colleagues used as the base stock a PAO blend that has really low traction coefficient along with an ester to solubilize the other additives, which is proprietary.
Based on earlier work in jet engines, Wedeven showed that this particular formulation could perform what he referred to as Run-In Polishing or RIP. After running a hypoid gear mesh for 32 hours, its roughness dropped by 20% and efficiency improved by one half to one percent.
“There are some innovative ideas coming out about this RIP notion. We’ve learned how to make chemistry smooth and protect surfaces,” Wedeven says.
Eventually though, he says, limits are reached—either the oil can’t take it or the surface can’t take it or the material below the surface can’t take it.
“So it’s a very complex phenomenon—where should the community go for improving efficiency? Do you put your money in with the oil or with the surfaces? Do both of them? There’s never a straight answer,” Wedeven says.
Speed agrees that it depends on the application.
“Different base oils from PAO to PAG to esters, they all have their pros and cons. And that’s the reality—everything has a pro and a con to it. Nothing is universally the best. The key thing,” Speed says, “is you have to embrace change. You’re not going to get greater efficiency by doing more of the same.”
REFERENCES
More information can be found at the following sources:
1.
How It Works: Driven Racing Oil. Available
here.
2.
Brown, S. (2015), “Base Oil Groups: Manufacture, Properties and Performance,” TLT,
71 (4), pp. 32-35. Available
here.
3.
Where the Energy Goes: Gasoline Vehicles. Available
here.
4.
Holmberg, et al. (2012) “Global energy consumption due to friction in passenger cars,”
Tribology International,
47 (March), pp. 221-234. Available
here.
5.
Synthetic Motor Oil - Do Synthetic Oils Really Work? Available
here.
6.
PAOs - ExxonMobil Chemical: Why sacrifice wear protection for energy efficiency? Available
here.
7.
Wedeven, L. (2018), “R.I.P. Gear Inefficiency,”
Lubes’N’Greases,
24 (11), pp. 28-33.
8.
Rudnick, L. (Ed.) and Rudnick, L. (Ed.). (2013),
Synthetics, Mineral Oils, and Bio-Based Lubricants: Chemistry and Technology, Second Edition, Boca Raton: CRC Press. Available
here.
Mary Beckman is a free-lance science writer based in Richland, Wash. You can contact her at mbeckman@nasw.org.