HIGHLIGHTS
•
A polyester copolymer prepared from a polyethylene glycol diester and a perfluoropolyether demonstrates potential as a sodium-ion battery solid electrolyte.
•
This reason for using the two starting materials is to take advantage of the exceptional chemical and thermal stability of the perfluoropolyether with the high solvation and charge mobilization properties of the polyethylene glycol diester.
•
Structural analysis indicates that the polyester copolymer is a biphasic material where the two phases become less distinct as the concentration of the dopant, NaTFSI, increases.
Research is continuing to identify and evaluate solid electrolytes in batteries because they exhibit less of a threat to fail due to a short-circuit which could potentially lead to a fire. But finding a solid electrolyte that can display comparable energy density and ionic conductivity compared to liquid electrolytes is still a work in progress.
In a previous TLT article,
1 a potential solid electrolyte was prepared through introduction of ionically conductive polymer additives into a ceramic matrix material by the use of cold sintering. The solid-state electrolyte used in this study is based in lithium, aluminum, titanium and phosphate, and is known as NASICON. In combination with a highly conductive poly (ionic liquid) gel, a high ionic conductivity in the range of 4.2 x 10
-4 Siemens per centimeter in a coin cell was achieved in the ceramic matrix material. The reason for this result is that interfacial resistance was reduced due to the uniform dispersion of the ionically conductive polymer through the ceramic matrix.
Polymer electrolytes have been under study for use in solid-state batteries because they are highly flexible, can be easily processed and offer favorable interfacial contacts with the electrodes. Michele Vittadello, director and professor of nanoscience at the Graduate Center, and professor of chemistry at the Medgar Evers College of the City University of New York, says, “Polymer electrolytes have been under evaluation for some time, but their potential has yet to be achieved. Initially, polymer electrolytes were mixed with solvent to form composite electrolyte gels. The concern with this approach is still flammability as battery manufacturers are seeking solvent free electrolytes.”
Of particular interest to Vittadello is to identify solid polymer electrolytes suitable for use in sodium-ion batteries. He says, “Sodium-ion batteries have potential for use particularly in stationary applications, which are growing in demand due to the increase in electricity consumption attributed to the construction of data centers.”
A significant number of polymers have been evaluated as hosts for polymer electrolytes. An appealing choice is polyethylene glycols (PEGs) because ethylene oxide monomer units in their backbones readily solvate sodium ions. In addition, these chains are highly mobile assisting with the ionic conductivity mechanism by imparting segmental motion to the charge carriers.
Perfluoropolyethers (PFPEs) are also under consideration as polymer hosts in solid electrolytes. Copolymers of PFPEs with PEGs containing the dopant NaTFSI displayed promise when used with the solvent DMSO that acts as a cryodepressant lowering the glass transition temperature of the polymer to facilitate ionic conductivity.
Vittadello and his colleagues have now synthesized a new sodium-ion battery solid electrolyte candidate that is based on a polyester copolymer.
Two conductivity channels
The polyester copolymer was prepared by the polymerization of a PEG diester and a PFPE. An enzymatic catalyst was used to facilitate the reaction. Vittadello says, “We decided to employ an enzymatic catalyst because of the advantages of precise structural control compared to conventional catalysts and also the ability to do the synthesis in one reaction vessel and to recover byproducts.”
The first step in the process was to convert a PEG diacid into a methyl ester through reaction with methanol in the presence of a few drops of concentrated sulfuric acid. Vittadello says, “PEG 600 diacid was used because this is the highest molecular weight member of this series, which is a liquid at room temperature.”
Once the methyl ester was produced, treatment with the PFPE at 90℃ under vacuum for 60 hours followed by filtration produced the polyester copolymer in the form of a semisolid gel at high yield. The researchers utilized the sodium salt of bis (trifluoromethane) sulfonimide at various concentrations, as a dopant, to enhance charge transport through the electrolyte.
Good sodium stability was observed with the polyester copolymer at room temperature. Vittadello says, “Our solid battery electrolyte candidate appears to function as a common mineral oil in protecting high reactive, sodium metal from oxidation.”
The polyester copolymer was characterized by several analytical techniques including elemental analysis, nuclear magnetic resonance, Fourier-transform infrared spectroscopy and impedance spectroscopy. Small angle X-ray scattering displayed evidence that the polyester copolymer consists of a biphasic material that contains one domain based on PFPE and PEG and a second domain based on PEG. As the concentration of dopant increases, the two phases become less distinct due to the dissolution of the dopant in both domains.
Ionic conductivity measurements showed a difference in performance between the two phases. The PEG phase exhibits ionic conductivity as high as 6.6 x 10
-5 Siemens per centimeter. In contrast, the value for the PFPE/PEG phase is 3.6 x 10
-5. The cationic transport numbers were measured to be above 0.80 and approaching unity, depending on the salt concentration.
Vittadello says, “Our intention in preparing the polyester copolymer is to combine the exceptional chemical and thermal stability of PFPE with the high solvation and charge mobilization properties of PEG. We consider the mechanism for ionic conductivity to be similar to cork-screw delocalization along the polymer axis. In this regard, sodium ions are coordinated with at least four oxygen atoms at any given time in these phases. The presence of the ester groups assists with intermolecular hopping of the sodium cations to other polymer chains. In the PFPE regions, the dopant assists with conductivity through interaction with difluoromethylene moieties present in the polymer backbone.”
An important parameter for using the polyester copolymer in stationary battery applications is thermal stability. Vittadello reports that both phases of this new stationary electrolyte are stable up to a temperature of 300℃.
Future work will involve synthesizing and evaluating additional polyester copolymers. Vittadello says, “We envision working with additional polyester copolymers not just as potential solid electrolytes but also as lubricant base stocks.”
Figure 3 contains an illustration of the components of the sodium-ion battery solid electrolyte and the potential for its use in batteries and lubricants.
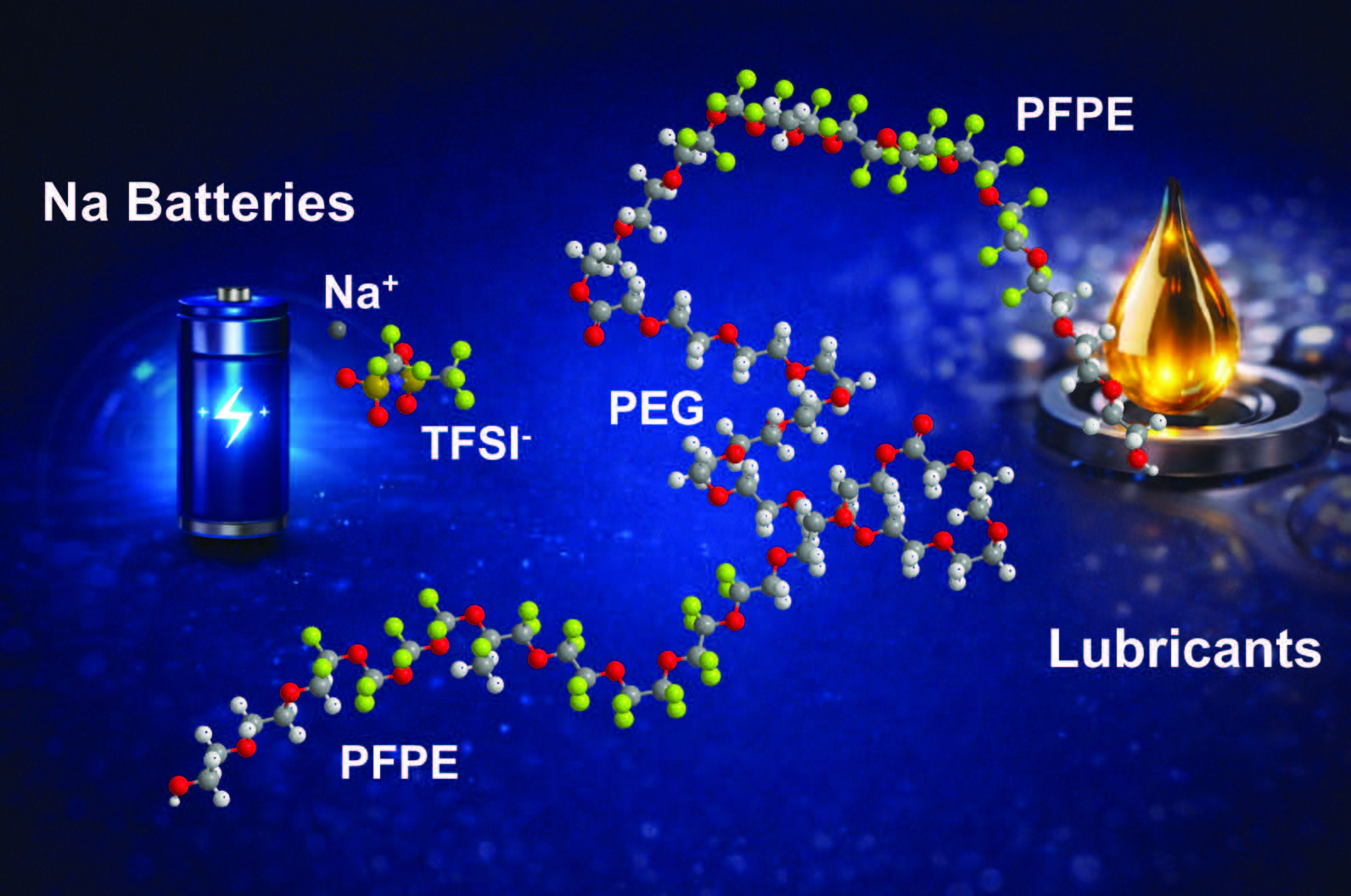 Figure 3. A polyester copolymer based on a polyethylene glycol (PEG) ester, perfluoropolyether (PFPE) and the dopant NaTFSI has potential for use as a sodium-ion battery solid electrolyte and in lubricants. Figure courtesy of the Medgar Evers College of the City University of New York.
Figure 3. A polyester copolymer based on a polyethylene glycol (PEG) ester, perfluoropolyether (PFPE) and the dopant NaTFSI has potential for use as a sodium-ion battery solid electrolyte and in lubricants. Figure courtesy of the Medgar Evers College of the City University of New York.
The National Science Foundation has recently awarded Vittadello’s team, including Professor Virinder Parmar, David Waxman, Adrian Tylim and Kumar Shivam, funds for exploring commercial applications of the electrolytes as battery materials and lubricants. Additional information can be found in a recent article
2 or by contacting Vittadello at
michele.vittadello89@login.cuny.edu.
REFERENCES
1.
Mills, A., Kalnaus, W., Su, Y., Williams, E., Zheng, X., Vaidyanathan, S., Hallinan, D., Nanda, J. and Yang, G. (2024), “Elucidating polymer binder entanglement in freestanding sulfide solid-state electrolyte membranes,”
ACS Energy Letters, 9 (6), pp. 2677-2684.