One approach to reduce stress on lithium-ion batteries is using porous electrode architectures.
A porous electrode lattice with an open octahedral structure was produced using 3D printing.
This lattice architecture enables a much higher percentage of the electrode to be used, leading to higher battery capacity and less stress.
Two of the most widely watched new technologies that may impact the lubricant industry are 3D printing (also known as additive manufacturing) and batteries. The former has the potential to be used in place of metal-removal operations while the latter may replace the internal combustion engine as a means to power automobiles.
New innovations in both of these areas have been the subject of articles in previous columns. For example, a previous TLT article discussed a new approach for 3D printing of aluminum alloys whose use is continuing to grow (
1). The researchers added nanoparticle grain refiners to facilitate the 3D printing of two widely used aluminum alloys, 6061 and 7075.
Printing of a zinc-based battery was discussed in a previous TLT article (
2). The technique used was screen printing that enabled the researchers to place a battery on a T-shirt.
Past work with lithium-ion batteries has shown that there are operating problems due to stress that can lead to such problems as flammability. Rahul Panat, associate professor of mechanical engineering at Carnegie Mellon University in Pittsburgh, says, “Conventional lithium ion battery electrodes have been prepared in block architecture. Only 30%-50% of the volume of the battery electrode is utilized, which leads to higher battery weight, slow charging and uneven stress distribution during charge/discharge cycles.”
One way to eliminate this issue is to design batteries using porous electrode architectures. Panat says, “The benefit of porous architecture is that more of the battery electrode is utilized, which enhances lithium ion transport and relieves stress on the battery as a result of uniform ingress of lithium into the electrode. The key is to increase the surface area of the electrode exposed to the lithium ions.”
Past attempts to develop porous architectures via 3D printing involved geometries similar to the fingers of two clasped hands according to Panat. He says, “This architecture can only be scaled up in a third dimension, height to increase the surface area. But the x-y plane is fixed, limiting porosity.”
A new porous lithium-ion battery electrode architecture recently was produced through the use of droplet-based 3D printing.
Open octahedral structure
Panat and his colleagues developed their method while utilizing the capabilities of the droplet-based method of Aerosol Jet 3D printing. He says, “In this process, nanoparticles are dispersed in liquid droplets that are a few microns in size through the use of pressurized air or ultrasonic energy and then deposited on a substrate.”
The researchers based the electrode on silver as a trial material as this element has a strong affinity for lithium. The silver nanoparticles were loaded into the ink at a concentration of 40%.
The base on which the electrode is formed is heated to 110 C. As the droplets reach the substrate, they coalesce and the solvents evaporate. Successive coalescence and evaporation processes leads to the formation of the porous microlattice structures. The process does not require any support material.
Panat indicated that there are two reasons for using 3D printing to produce the battery electrode. He says, “3D printing enables us to work with geometries that are impossible to make by more traditional methods. We can also work with an array of materials in combinations that are difficult to use otherwise.”
The researchers developed a porous lattice with an open octahedral structure. Says Panat: “We deliberately designed the porous lattice for this specific geometry due to its large number of degrees of freedom compared to other geometries. Open octahedral structures exhibit four degrees of freedom and can be produced within the constraints of the 3D printer. The high number of connections (eight at every point of the porous lattice) leaves room for one or two of the connections to break without impacting the performance of the electrode.”
Placement of five by five by five individual units of lattice generates the lattice shown in Figure 2. Lithiation of the porous lattice led to a much higher utilization of the lattice compared to a conventional dense block structure for a given time of charging.
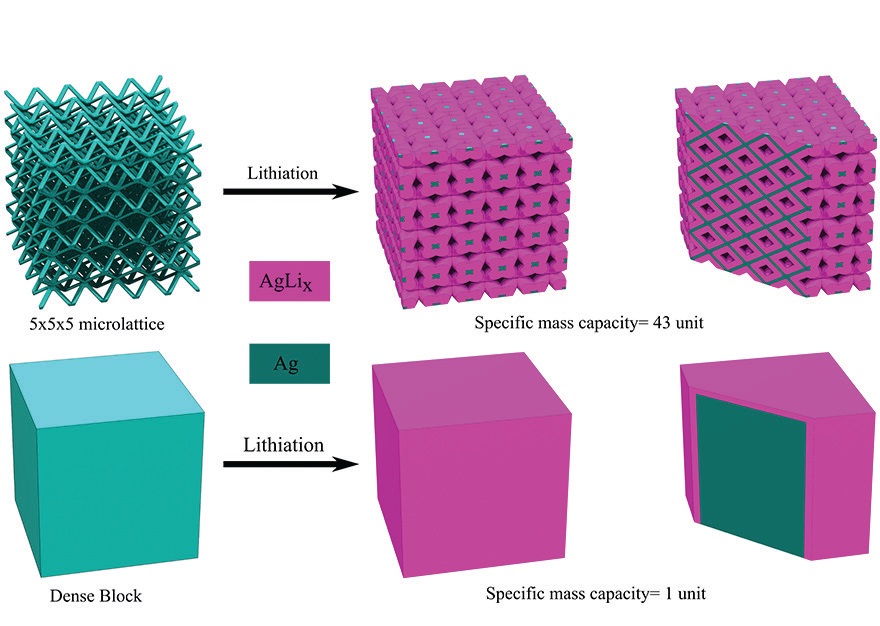 Figure 2. A lithium-ion battery electrode prepared with a porous lattice (on top) through the use of 3D printing exhibits enhanced ion transport because more of its surface is exposed to the electrode compared to a dense block electrode (on the bottom). (Figure courtesy of Carnegie Mellon University.)
Figure 2. A lithium-ion battery electrode prepared with a porous lattice (on top) through the use of 3D printing exhibits enhanced ion transport because more of its surface is exposed to the electrode compared to a dense block electrode (on the bottom). (Figure courtesy of Carnegie Mellon University.)
The surface area to volume of the porous lattice electrode is approximately 0.5, which is significantly higher than the figure for a dense block electrode that approaches zero as the electrode size increases. Panat says, “Engagement of a much higher percentage of the electrode leads to a higher battery capacity and a reduction in stress.”
The researchers produced porous electrodes with surface areas between 1 and 2 square millimeters and a height of 0.5 millimeter. Evaluation of a lithium-ion battery with the porous electrodes is at a preliminary stage. “We have evaluated the porous electrodes for up to 40 charge/discharge cycles with continuing strong performance gains,” Panat says. “Future work will involve evaluating the reliability and durability of the battery over a longer operating period.”
Additional information on this research can be found in a recent article (
3) or by contacting Panat at
rpanat@andrew.cmu.edu.
REFERENCES
1.
Canter, N. (2018), “Manufacturing of aluminum alloys by 3D printing,” TLT,
74 (1), pp. 14-15.
2.
Canter, N. (2017), “Printing batteries,” TLT,
73 (8), pp. 10-11.
3.
Saleh, M., Li, J., Park, J. and Panat, R. (2018), “3D printed hierarchically-porous microlattice electrode materials for exceptionally high specific capacity and areal capacity lithium ion batteries,
Additive Manufacturing,
23, pp. 70-78.