Metathesis powers marketability of biobased lubricants
Jeanna Van Rensselar, Contributing Editor | TLT Feature Article November 2013
The best of both worlds—biobased eco-friendliness with the performance of synthetics.
KEY CONCEPTS
• Metathesized biobased oils could be the key to developing biobased, environmentally acceptable lubricants that also perform well.
• Metathesized base oils perform differently than conventional base oils, reducing the additives needed to boost performance in biobased fuels and lubricants.
• The full-scale production of metathesized biobased products is underway with lubricants derived from these building blocks currently being introduced into the market.
LUBRICANT RESEARCHERS NEED NEW TOOLS that provide high viscosity index, low volatility, low-temperature fluidity, high-temperature performance and good oxidative stability for today’s demanding lubricant needs. Experts think that metathesis-processed renewable oils will be the key to producing high-performance, biobased base oils and additives.
Dr. Kathleen O’Leary Havelka, director, product development for Elevance Renewable Sciences, Inc., in Woodridge, Ill., says, “Looking forward to 2020, the critical properties that will be needed are low-temperature fluidity, improved viscometrics, lower volatility, clean operation and high-temperature oxidative resistance. As the industry progresses to even smaller displacement engines with higher power densities and higher temperatures with turbochargers, all while improving fuel economy, it is critically important that these lubricant performance properties are improved to maintain equipment life.”
The metathesis process brings the ability to efficiently make olefins and difunctional (1) specialty chemicals from renewable feedstocks; enabling, for the first time, biobased fluids as practical, high performance and sustainable substitutes for synthetic lubricants.
With metathesis, biobased lubricants, including renewable green PAOs, are possible and can perform as well as PAOs produced from petroleum and other synthetic fluids. Not only does this improve marketability, it addresses one of the historical objections to biobased lubricants: biobased materials cannot offer top performance.
WHAT IS METATHESIS?
Metathesis is derived from the Greek words meta (change) and thesis (position). As with other chemical reactions during metathesis, the existing bonds between atoms are broken and new bonds are formed. In olefin metathesis (2), the carbon-carbon double bonds are broken and the resulting fragments change places with one another. For example, the olefin metathesis of propene results in the formation of butene and ethene.
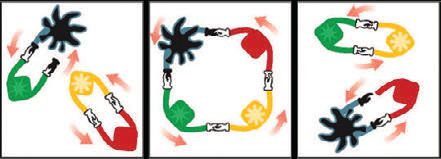
The metathesis mechanism. (Photo courtesy of Lou Honary, National Ag-Based Lubricants Center at the University of Northern Iowa)
HISTORY
Early research led to the ability to form carbon chains with double bonds (olefins) and also convert propene into butene and ethene by introducing a mixture of tri-isobutyl aluminum and molybdenum oxide to aluminum oxide.
The connection between these two discoveries was made in the 1950s by Dr. Nissim Calderon at Goodyear. Calderon showed that the same type of reaction took place in both processes and called the reaction “olefin metathesis.”
Since Calderon’s discovery, substances were produced through metathesis, even though no one really understood how the catalyst worked. Further breakthroughs came over the next few decades from scientists Yves Chauvin, Robert H. Grubbs and Richard R. Schrock. Chauvin and his student, Jean-Louis Hérrison, proposed that the catalyst was a metal carbine (3); now known as metal alkylidene. Chauvin also showed how the metal compound functions as a catalyst in metathesis.
Chauvin’s mechanism is often compared to a dance involving the metal alkylidene pair of the catalyst and the carbon-carbon pair of an alkene. Each partner of the catalyst pair has two hands and holds both hands of the other. When the catalyst pair meets the alkene pair, the two pairs unite, forming a square. Then they let go of each other’s hands, leave their old partners and dance on with their new partners. The new metal-alkylidene catalyst pair will then unite with another dancing alkene pair, forming a new square, and continue the metathesis process. In this way, Chauvin showed the efficiency of metal-alkylidene catalysts in the metathesis reaction.
With the mystery of the catalyst’s function gone, the remaining problem was that none of the known well-defined metal alkylidenes acted as catalysts in all-important olefin metathesis. Olefin metathesis required stable, well-defined catalysts that reacted only with double bonds and did not affect other parts of the molecules. In addition, the reactivity needed the ability to be adjusted to the purpose.
In the 1970s, Schrock built on the work of Chauvin by researching new alkylidene complexes. He gradually developed an understanding of what metals could be used in the catalysts and how they functioned. Molybdenum and tungsten appeared to perform best.
There was still uncertainty about which groups would bind to the metal and yield stable, active alkylidene compounds. In 1990, Schrock and his coworkers reported the discovery of a group of very active, well-defined molybdenum catalysts. Researchers then began to grasp how olefin metathesis could significantly improve and replace a number of traditional, less efficient organic synthesis methods.
In 1992, Grubbs and his coworkers discovered a catalyst with the metal ruthenium. It was stable in air and demonstrated higher selectivity but lower reactivity than Schrock’s catalysts. The new catalyst also had the ability to initiate metathesis in the presence of alcohols, water and carboxyl acids. Grubbs continued to improve his catalysts, which eventually became the first well-defined catalysts for general metathesis processes. Catalyst 2 was named the Grubbs Catalyst™ and has become the standard with which all new catalysts are still compared.
The contributions of Chauvin, Grubbs and Schrock are transforming the development and production of a number of substances including chemicals, rubber and life-saving pharmaceuticals. Their research opened up opportunities to take full advantage of the diversity of organic molecules. For their work, all three scientists earned a Nobel Prize for chemistry in 2005.
Subsequent researchers made important contributions and are continuing the development of new metathesis catalysts designed to solve specific problems such as the synthesis of complex natural substances.
Considering the relatively short time Schrock’s and Grubbs’ catalysts have been available, a remarkable breadth of applications have developed, including additives for polymers, pheromones and specialty chemicals. Today catalytic metathesis results in more end products and fewer waste products, which means cleaner, more environmentally friendly production.
THE BENEFITS OF OLEFIN METATHESIS (4)
There are three basic reasons why olefin metathesis has valuable real-world applications in a broad array of fields:
• It’s an eco-friendly process. Because olefin metathesis is promoted by a catalyst, a well-designed process will generate almost no unusable byproducts.
• It’s cost-effective. The same factors that make olefin metathesis a green process also make it cost-effective.
• It’s easily scalable. Olefin metathesis reactions have been successfully performed in scales ranging from milligrams to several tons.
METATHESIS AND LUBRICANTS
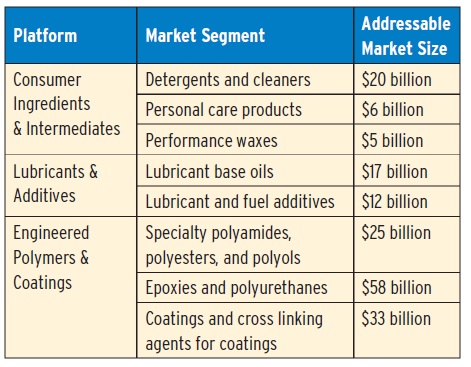
Metathesis-processed renewable oils have broad application in many large and growing markets, including lubricants. The following table shows the estimated market size for each (5).
Historically, one of the major deterrents to the widespread adoption of biobased lubricants is the cost of production; specifically that cost has been high relative to petrochemical- based lubricants. Reducing conversion, capital and energy costs is exactly where metathesis enabled conversions can be a game changer.
Another large historical deterrent to wider acceptance is the inferior performance of biobased lubricants (relative to synthetic lubricants). The unsaturation present in vegetable oil gives it poor oxidative stability. However, metathesis reacts at these sites of unsaturation, transforming the unsaturated oil into a consistent and stable building block that can be converted into novel lubricants.
Havelka says, “The difunctional reaction products of metathesis-processed natural oils give us versatile chemical building blocks that we can react further. This enables new structures that allow enhanced base oil and lubricant additive performance. These transformations cannot be done easily with crude oil or simple olefin-based materials and technologies. Elevance’s difunctional materials give us the ability to create novel molecular architecture such as controlling molecular weight, geometry and degree of branching. This creates new ways to manipulate lubricant properties.”
“Lubricant viscometric behavior at cold startup is an important variable relative to fuel economy and engine protection. One important strategy to improve fuel economy and low-temperature performance has been to reduce bulk viscosity. Reducing viscosity, however, can have a detrimental effect on wear and friction at operating temperatures,” Havelka says. “From a formulation point of view, the industry has used a number of additives, such as antiwear, friction modifiers, antioxidants, dispersants, detergents and viscosity modifiers to balance engine performance requirements in motor oils. These legacy technologies have contributed to enabling lubricant flow at lower temperatures, multigrade motor oils that function across a broad operating temperature range, but more improvement is needed for more severe operating conditions that are emerging.”
MATERIA’S METATHESIS CATALYSTS (6)
Materia commercialized the Nobel Prizewinning olefin metathesis catalyst technology discovered at the California Institute of Technology (Caltech) by professor Robert H. Grubbs. The Grubbs Catalyst™ technology is a ruthenium-based chemical catalyst system that can be employed in most olefin-based applications. When compared to traditional processes, Grubbs Catalyst products usually offer significant reductions in the number of manufacturing steps and the amount of byproducts, solvents and waste.
In 2003, Cargill worked with Materia to explore how metathesis might be applied to the synthesis of high-value products from natural oils. In 2007, the venture was spun off with independent financing from the private equity firm, TPG, creating Elevance Renewable Sciences.
Elevance’s modified biobased materials offer the possibility of a wide variety of chain lengths and multiple functional groups. These can be further tailored for unique geometries, improved viscosity index and low-temperature fluidity. This flexibility allows the development of biobased products for a wide range of base oil and lubricant additive applications.
THE ELEVANCE BIOREFINERY
Woodridge, Ill.-based, Elevance Renewable Sciences, Inc., recently announced commercial shipments from its first worldscale biorefinery located in Gresik, Indonesia (a joint venture with Wilmar International Limited). The biorefinery is the first based on Elevance’s proprietary metathesis technology.
The facility produces novel specialty chemicals (including multifunctional esters such as 9-decenoic methyl ester), a unique distribution of biobased alpha and internal olefins, which includes decene and a premium mixture of oleochemicals. It has a capacity of 180 kMT (approximately 400 million pounds) with the ability to expand up to 360 kMT (approximately 800 million pounds) of products.
The high-value performance specialty chemicals available include Inherent C18 Diacid, a mid-chain length, biobased diacid that enables producers of polyamides, polyurethanes, lubricants and adhesives to significantly expand their portfolios with cost-competitive products that exceed the performance of shorter-chain diacids.
Elevance’s process to produce C18 diacid (also known as octadecanedioic acid or ODDA) uses materials produced from this metathesis- based biorefinery in Gresik, Indonesia. The process allows Elevance to make Inherent C18 Diacid with the purity required for applications like polymers.
As a diacid (or if converted to diamine), a C18 diacid makes possible several new base polymers that can result in more than 100 new compounds or formulations. For example, Inherent C18 Diacid will allow polyamides to enter new automotive and electronic applications that demand better hydrolytic performance, improved optical properties and greater material toughness or flexibility. In polyester polyols, C18 diacid makes possible new, previously unattainable prepolymers, helping polyurethane manufacturers create polymers with exceptional solvent resistance, hydrolytic stability, optical clarity and toughness that will benefit automotive applications.

Elevance-Wilmar Joint Venture Biorefinery, Gresik, Indonesia. (Photo courtesy of Elevance Renewable Sciences, Inc.)
Havelka also sees the possibility for metathesis-processed natural oil derivatives to be used in greases. The possibilities range from a bio-derived base oil to materials that help to make additives compatible, offering the possibility of more efficient grease production and quality.
Lou Honary, professor and director of the National Ag-Based Lubricants Center at the University of Northern Iowa in Waterloo, Iowa, says, “The real advantage of metathesis is that more tailor-made base oils can be designed for more sophisticated lubricants and greases.”
ENVIRONMENTAL ADVANTAGES
Dr. Susan Gardner, regional R&D chemist for Klüber Lubrication North America L.P. in Londonderry, N.H., explains that finding the sweet spot between performance and environmental safety is tough. “One of the biggest challenges formulators face is finding that balance between performance and eco-friendliness. Unfortunately, some of the best-performing raw materials are just not safe for the environment.”
Metathesis could be the answer.
There are a few reasons why metathesis of natural oils is a more environmentally friendly procedure: (1.) the primary raw materials are from renewable resources, (2.) the metathesis catalysts enable a relatively low-pressure, low-temperature process that consumes significantly less energy and reduces greenhouse gas emissions compared to petrochemical technologies, while generating less waste and fewer byproducts and (3.) the process may impart qualities such as better viscosity and oxidative stability when properly formulated.
The metathesis catalysts developed by Schrock and Grubbs make biobased feedstocks practical for the chemical industry. Multiple renewable oils such as palm, mustard, soybean and, when they become commercially available, jatropha and algal oils are viable feedstocks for conversion into specialty chemical-building blocks that can be processed further into functional monomers, polymers, surfactants and high-performing biobased and biodegradable lubricants.
Elevance and its partner, Wilmar International, recently began shipping commercial specialty products from their biorefinery in Gresik, Indonesia. These renewable specialty products will be processed into a number of products including lubricant base oils with improved viscometrics and fuel economy, as well as additives for lubricants and fuels.
Between the environmental advantages and the performance advantages, Honary believes that metathesis will improve the marketability of biobased lubricants. “While for the last 25 years we have been working on biobased lubes and greases using vegetable oils, I consider such efforts as transitional toward more universal biobased products,” he explains. “Vegetable oils cannot provide the uniformity of performance that current petroleum base oils do because changes in the growing conditions impact the properties of the seeds that provide the oil. Now products such as estolides, complex esters and metathesis-processed oils can be made from renewable basestocks, including vegetable oils, but offer uniformity and consistency that is similar to petroleum base oils. This means that the odds of biobased lubricants and greases becoming commonly used in all fields will increase dramatically.”
Havelka concludes, “Looking forward, we expect requirements will become ever more stringent for low-temperature flow and wear protection. It is difficult to balance low-temperature flow while maintaining film strength and thickness at higher operating temperatures. Our new generation biobased chemistry enables more diverse functionality tailored to offer new solution and surface performance. This more diverse set of options gives biobased lubricant formulators more tools to tailor material characteristics, such as traction coefficient and viscosity index, to specific problems.”
ALL THINGS METATHESIS
Everything anyone would ever want to know about metathesis is at the All Things Metathesis blog: http://allthingsmetathesis.com/. The blog is a resource for olefin metathesis and provides a setting for metathesis users to brainstorm. From the blog site, visitors can register for the All Things Metathesis RSS feed and subscribe to All Things Metathesis emails.
The blog was developed by Pasadena, Calif.-based Materia, Inc., to establish a resource on olefin metathesis and a forum for the growing number of metathesis enthusiasts. Primary contributor Andy Nickel says he became interested in metathesis after he skipped band practice one day to attend a graduate-student literature seminar called, “Ruthenium-Catalyzed Olefin Metathesis.” He went on to earn his doctorate in natural product synthesis from Yale University in 2005. After working as a medicinal chemist for three years, he joined Materia in the summer of 2008.
Honary adds, “If those working with this technology can deliver consistent products at a cost that is close to that of high-oleic vegetable oils, then, in my view, they have a clear path to success. There will be a solid market for tailor-made renewable base oils that can meet the extreme performance conditions of industrial and automotive lubes and greases. Metathesis could possibly deliver that.”
REFERENCES
1. The term difunctional is defined as two reactive sites in each compound molecule.
2. Metathesis frequently refers to olefin metathesis.
3. A metal carbene is a compound in which the metal is bound to the carbon with a double bond.
4. From here.
5. From here.
6. From here.

Jeanna Van Rensselar heads her own communication/public relations firm, Smart PR Communications, in Naperville, Ill. You can reach her at jeanna@smartprcommunications.com.